MELTS¶
Versions of MELTS implemented are:
- MELTS v. 1.0.2 ➞ (rhyolite-MELTS, Gualda et al., 2012)
- MELTS v. 1.1.0 ➞ (rhyolite-MELTS + new CO2, works at the ternary
minimum)
- MELTS v. 1.2.0 ➞ (rhyolite-MELTS + new H2O + new CO2)
- pMELTS v. 5.6.1
Initialize tools and packages that are required to execute this notebook.¶
from thermoengine import equilibrate
import matplotlib.pyplot as plt
import numpy as np
%matplotlib inline
Create a MELTS v 1.0.2 instance.¶
Rhyolite-MELTS version 1.0.2 is the default model.
melts = equilibrate.MELTSmodel()
Optional: Generate some information about the implemented model.¶
oxides = melts.get_oxide_names()
phases = melts.get_phase_names()
#print (oxides)
#print (phases)
Required: Input initial composition of the system (liquid), in wt% or grams of oxides.¶
Early Bishop Tuff average melt inlusion composition
feasible = melts.set_bulk_composition({'SiO2': 77.5,
'TiO2': 0.08,
'Al2O3': 12.5,
'Fe2O3': 0.207,
'Cr2O3': 0.0,
'FeO': 0.473,
'MnO': 0.0,
'MgO': 0.03,
'NiO': 0.0,
'CoO': 0.0,
'CaO': 0.43,
'Na2O': 3.98,
'K2O': 4.88,
'P2O5': 0.0,
'H2O': 5.5})
Optional: Suppress phases that are not required in the simulation.¶
b = melts.get_phase_inclusion_status()
melts.set_phase_inclusion_status({'Nepheline':False, 'OrthoOxide':False})
a = melts.get_phase_inclusion_status()
for phase in b.keys():
if b[phase] != a[phase]:
print ("{0:<15s} Before: {1:<5s} After: {2:<5s}".format(phase, repr(b[phase]), repr(a[phase])))
Nepheline Before: True After: False
OrthoOxide Before: True After: False
Compute the equilibrium state at some specified T (°C) and P (MPa).¶
Print status of the calculation.
output = melts.equilibrate_tp(760.0, 175.0, initialize=True)
(status, t, p, xmlout) = output[0]
print (status, t, p)
success, Minimal energy computed. 760.0 175.0
Summary output of equilibrium state …¶
melts.output_summary(xmlout)
T (°C) 760.00
P (MPa) 175.00
Quartz 1.2006 (g) SiO2
Liquid 101.5218 (g) wt %:SiO2 73.63 TiO2 0.08 Al2O3 11.87 Fe2O3 0.20 Cr2O3 0.00 FeO 0.47 MnO 0.00 MgO
0.03 NiO 0.00 CoO 0.00 CaO 0.42 Na2O 3.79 K2O 4.61 P2O5 0.00 H2O 4.90
Sanidine 2.3292 (g) K0.49Na0.50Ca0.01Al1.01Si2.99O8
Water 0.5285 (g) H2O
Output thermodynamic properties of any phase present in the system¶
… or the sum of all phases in the system
props = melts.get_list_of_properties()
for prop in props:
print ("{0:<20s} {1:13.6e} {2:<10s}".format(prop, melts.get_property_of_phase(xmlout,'System', prop), \
melts.get_units_of_property(prop)))
Mass 1.055800e+02 g
GibbsFreeEnergy -1.723511e+06 J
Enthalpy -1.444769e+06 J
Entropy 2.697990e+02 J/K
HeatCapacity 1.419994e+02 J/K
DcpDt 1.086318e-02 J/K^2
Volume 4.885928e+00 J/bar
DvDt 8.637431e-04 J/bar-K
DvDp -1.354787e-04 J/bar^2
D2vDt2 3.084601e-07 J/bar-K^2
D2vDtDp -2.083152e-07 J/bar^2-K
D2vDp2 8.811872e-08 J/bar^3
Density 2.160900e+00 g/cm^3
Alpha 1.767818e-04 1/K
Beta 2.772834e-05 1/bar
K 3.606418e+00 GPa
K' 2.245704e+01 none
Gamma 4.677198e-02 none
Output chemical affinities and potential compositions of undersaturated phases¶
dict = melts.get_dictionary_of_affinities(xmlout, sort=True)
for phase in dict:
(affinity, formulae) = dict[phase]
print ("{0:<20s} {1:10.2f} {2:<60s}".format(phase, affinity, formulae))
Leucite 1843.53 K0.50Na0.50AlSi2O5.50(OH)1.00
Tridymite 2141.04 SiO2
Cristobalite 2204.71 SiO2
Spinel 3443.41 Fe''1.00Mg0.00Fe'''0.00Al1.00Cr1.00Ti0.00O4
Magnetite 6100.91 Fe3O4
Coesite 6702.07 SiO2
Ilmenite ss 7040.63 Mn0.00Fe''0.00Mg0.50Fe'''1.00Al0.00Ti0.50O3
Orthopyroxene 7481.78 Na0.00Ca0.50Fe''0.00Mg1.50Fe'''0.00Ti0.00Al0.00Si2.00O6
Pigeonite 7899.03 Na0.00Ca0.50Fe''0.00Mg1.50Fe'''0.00Ti0.00Al0.00Si2.00O6
Olivine 8602.61 (Ca0.00Mg0.00Fe''0.50Mn0.50Co0.00Ni0.00)2SiO4
Fayalite 10535.94 Fe2SiO4
Ilmenite 10912.99 FeTiO3
Rutile 13232.93 TiO2
Sillimanite 14224.20 Al2SiO5
Andalusite 14583.54 Al2SiO5
Corundum 14605.08 Al2O3
Kyanite 18244.50 Al2SiO5
Garnet 18772.08 (Ca0.50Fe''0.50Mg0.00)3Al2Si3O12
Hematite 18830.66 Fe2O3
Biotite 19819.30 K(Fe''0.50Mg0.50)3AlSi3O10(OH)2
Nepheline 20015.65 NaAlSiO4
Sphene 24104.31 CaTiSiO5
Muscovite 25188.37 KAl2Si3AlO10(OH)2
Phlogopite 28723.51 KMg3AlSi3O10(OH)2
Forsterite 29210.52 Mg2SiO4
OrthoOxide 29553.98 Fe''0.50Mg0.00Fe'''1.00Ti1.50O5
Kalsilite 33671.36 KAlSiO4
Cummingtonite 37212.14 Ca0.00Fe0.00Mg7.00Si8O22(OH)2
Perovskite 38528.62 CaTiO3
Anthophyllite 39317.80 Ca0.00Fe0.00Mg7.00Si8O22(OH)2
Periclase 43143.46 MgO
Nepheline ss 43681.21 Na2.00K2.00Ca0.00[]0.00Al4.00Si4.00O16
Melilite 48421.67 Na0.00Ca2.00Al2.00Mg-0.00Fe0.00Si1.00O7
Kalsilite ss 51793.54 Na2.00K2.00Ca0.00[]0.00Al4.00Si4.00O16
Hornblende 54565.30 NaCa2Mg2.00Fe2+2.00Al1.00Fe3+0.00Al2Si6O22(OH)2
Aenigmatite 58029.65 Na2Fe5TiSi6O20
Solid Alloy 69174.78 Fe1.00Ni0.00
Liquid Alloy 73771.78 Fe1.00Ni0.00
Akermanite 80666.47 Ca2MgSi2O7
Gehlenite 84296.04 Ca2Al2SiO7
Lime 113534.98 CaO
Aegirine 141581.81 NaFeSi2O6
Actinolite 999999.00 Ca0.00Fe3.50Mg3.50Si8O22(OH)2
Whitlockite 999999.00 Ca3(PO4)2
Apatite 999999.00 Ca5(PO4)3OH
Chromite 999999.00 FeCr2O4
Plagioclase 999999.00 K0.00Na0.50Ca0.50Al1.50Si2.50O8
Augite 999999.00 Na0.00Ca0.50Fe''0.00Mg1.50Fe'''0.00Ti0.00Al0.00Si2.00O6
Titanaugite 999999.00 Na0.00Ca0.50Fe''0.00Mg1.50Fe'''0.00Ti0.00Al0.00Si2.00O6
Panunzite 999999.00 Na2.00K2.00Ca0.00[]0.00Al4.00Si4.00O16
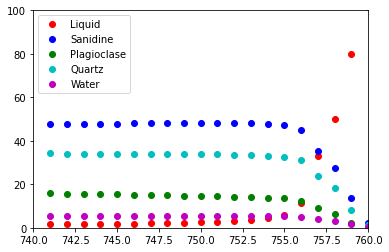
Run the sequence of calculations along a T, P=constant path:¶
Output is sent to an Excel file and plotted in the notebook
number_of_steps = 20
t_increment_of_steps = -1.0
p_increment_of_steps = 0.0
plotPhases = ['Liquid', 'Sanidine', 'Plagioclase', 'Quartz', 'Water']
# matplotlib colors b : blue, g : green, r : red, c : cyan, m : magenta, y : yellow, k : black, w : white.
plotColors = [ 'ro', 'bo', 'go', 'co', 'mo']
wb = melts.start_excel_workbook_with_sheet_name(sheetName="Summary")
melts.update_excel_workbook(wb, xmlout)
n = len(plotPhases)
xPlot = np.zeros(number_of_steps+1)
yPlot = np.zeros((n, number_of_steps+1))
xPlot[0] = t
for i in range (0, n):
yPlot[i][0] = melts.get_property_of_phase(xmlout, plotPhases[i])
plt.ion()
fig = plt.figure()
ax = fig.add_subplot(111)
ax.set_xlim([min(t, t+t_increment_of_steps*number_of_steps), max(t, t+t_increment_of_steps*number_of_steps)])
ax.set_ylim([0., 100.])
graphs = []
for i in range (0, n):
graphs.append(ax.plot(xPlot, yPlot[i], plotColors[i]))
handle = []
for (graph,) in graphs:
handle.append(graph)
ax.legend(handle, plotPhases, loc='upper left')
for i in range (1, number_of_steps):
output = melts.equilibrate_tp(t+t_increment_of_steps, p+p_increment_of_steps)
(status, t, p, xmlout) = output[0]
print ("{0:<30s} {1:8.2f} {2:8.2f}".format(status, t, p))
xPlot[i] = t
for j in range (0, n):
yPlot[j][i] = melts.get_property_of_phase(xmlout, plotPhases[j])
j = 0
for (graph,) in graphs:
graph.set_xdata(xPlot)
graph.set_ydata(yPlot[j])
j = j + 1
fig.canvas.draw()
melts.update_excel_workbook(wb, xmlout)
melts.write_excel_workbook(wb, "MELTSv102summary.xlsx")
success, Minimal energy computed. 759.00 175.00
success, Minimal energy computed. 758.00 175.00
success, Minimal energy computed. 757.00 175.00
success, Minimal energy computed. 756.00 175.00
success, Minimal energy computed. 755.00 175.00
success, Minimal energy computed. 754.00 175.00
success, Minimal energy computed. 753.00 175.00
success, Minimal energy computed. 752.00 175.00
success, Minimal energy computed. 751.00 175.00
success, Minimal energy computed. 750.00 175.00
success, Minimal energy computed. 749.00 175.00
success, Minimal energy computed. 748.00 175.00
success, Minimal energy computed. 747.00 175.00
success, Minimal energy computed. 746.00 175.00
success, Minimal energy computed. 745.00 175.00
success, Minimal energy computed. 744.00 175.00
success, Minimal energy computed. 743.00 175.00
success, Optimal residual norm. 742.00 175.00
success, Minimal energy computed. 741.00 175.00